
Belliveau, J., & Papoutsakis, E. T. (2022). Extracellular vesicles facilitate large-scale dynamic exchange of proteins and RNA among cultured Chinese hamster ovary and human cells. Biotechnology and bioengineering, 10.1002/bit.28053. Advance online publication. https://doi.org/10.1002/bit.28053
Cover Photo: The cover image is based on the Article Extracellular vesicles facilitate large-scale dynamic exchange of proteins and RNA among cultured Chinese hamster ovary and human cells by Jessica Belliveau and Eleftherios T. Papoutsakis.

J Wyffels et al. 2020. Testosterone and semen seasonality for the sand tiger shark Carcharias taurus. Biology of Reproduction 10:.4, 876-887. https://doi.org/10.1093/biolre/ioz221
Cover Photo: The cover image is based on the Article Testosterone and semen seasonality for the sand tiger shark Carcharias taurus by J Wyffels et al. (2020). https://doi.org/10.1093/biolre/ioz221

Cody E. Garrison, Kyra A. Price, Erin K. Field. 2019. Environmental Evidence for and Genomic Insight into the Preference of Iron-Oxidizing Bacteria for More-Corrosion-Resistant Stainless Steel at Higher Salinities. Applied and Environmental Microbiology. 85(14) e00483-19; DOI: 10.1128/AEM.00483-19
Cover Photo: Scanning electron microscope image of a novel species of iron-oxidizing bacteria, Mariprofundus erugo, isolated from stainless steel deployed in a North Carolina estuarine river. Iron-oxidizing bacteria are one of the initial colonizing microorganisms during biocorrosion of steel, and this new species may be adapted to more-advanced anthropogenic iron sources such as stainless steel. Growth is characterized by helical stalk iron oxide morphologies as seen here.

Spaulding, HR, Kelly, EM, Quindry JC, Sheffield, JB, Hudson MB, Selsby JT. 2018. Autophagic dysfunction and autophagosome escape in the mdx mus musculus model of Duchenne muscular dystrophy. Acta Physiologica. 222(2):690-703. doi: 10.1111/apha.12944.
Cover Photo: Autophagosome and lysosome colocalization. Confocal images at 639 magnification of DAPI (blue), p62 (green) and Lamp2 (red) from 14-mo-old dystrophic and control mice diaphragms.Acta Physiol (Oxf)

Percak-Dennett E, He S, Converse B, Konishi H, Xu H, Corcoran A, Noguera D, Chan C, Bhattacharyya A, Borch T, Boyd E, Roden EE. 2017. Microbial acceleration of aerobic pyrite oxidation at circumneutral pH. Geobiology. (5):690-703. doi: 10.1111/gbi.12241. Epub 2017 Apr 27.
Cover Photo: Cryo-SEM image demonstrating the intimate cell-mineral association of an aerobic pyrite-oxidizing enrichment culture after ca. 45 days of growth. Cells are shown in false orange color. Micron-size pyrite particles are aggregates of numerous small framboidal crystallites.
2016. Correlative Microscopy Supplement. Microscopy and Microanalysis. Issue 143

Polenova, T., Gupta, R., Goldbourt, A. 2015. Magic Angle Spinning NMR Spectroscopy: A Versatile Technique for Structural and Dynamic Analysis of Solid-Phase Systems. Anal. Chem. 87 (11): 5458-5469.

Al-Hinai, M.A., Jones, S.W., Papoutsakis, E.T. 2015. The Clostridium sporulation programs: diversity and preservation of endospore differentiation. Microbiol Mol Biol Rev doi:10.1128/MMBR.00025-14.
Cover Photo: Transmission electron micrographs (TEMs) of Clostridium acetobutylicum at different stages of its developmental (sporulation) program, from a symmetrically dividing vegetative cell on the left to a maturing spore on the right. Pseudocolor is used to accentuate key features of the various cellular forms, notably those of granulose vesicles (light green), the developing endospore (light blue), the spore membrane (dark yellow), and the spore cortex (brown), which is surrounded by the thin “black” spore coat. The phenotypic characteristics of the sporulation program of Clostridium organisms are similar to that of Bacillus spp., with the key exception that Clostridium organisms synthesize the storage material granulose (made of amylopectin). Granulose is packed in vesicles, whose accumulation leads to swelling of the cell, giving rise to the cigar-shaped clostridial cell form that looks like a spindle. The genus name “Clostridium” (as well as the class name “Clostridia”) derives from the Greek word for spindle, klōstēr. Despite the similar phenotypic characteristics, the molecular orchestrations of sporulation are quite distinct between Clostridium and Bacillus. TEM images are not to scale and were arranged for artistic impact. TEM images are from the collection of the Papoutsakis lab at the University of Delaware and were obtained at the Delaware Biotechnology Institute BioImaging Center. The cover composition was conceived by E. Terry Papoutsakis and executed by Patrick Lane of ScEYEnce Studios.
Caplan, J.L., Kumar, A. S., Park, E., Pasmanabhan, M. S., Hoban, K., Modla, S., Czymmek, K., Dinesh,-Kumar, S. P. 2015. Chloroplast stromules function during innate immunity. 34 (1): 45-57.
Cover Photo: The 3D-rendered image shows tubular projections called stromules from multiple chloroplasts (blue) surrounding a nucleus (yellow) during a plant immune response, observed by confocal laser scanning microscopy. Chloroplasts (magenta) in the mesophyll cells are in the background. Defense signaling molecules and proteins from chloroplasts may be transported to the nucleus via such stromules.
Naik, M. U., Caplan, J. L., Naik U. P. 2015. Junctional adhesion molecule-A suppresses platelet integrin αIIbβ3 signaling by recruiting Csk to the integrin-c–Src complex. Blood. 123 (9): 1393-1402.
Cover Photo: Immunolocalization of JAM-A (red) and integrin αIIbβ3 (blue) on human platelet exposed to immobilized fibrinogen. Platelets were also stained with phalloidin to visualize F-actin (green). The image is a 3D rendering of a super-resolution structured illumination microscopy (SIM) dataset.

Roberson, A., Spence, C., Bais, H. P. 2014. Belowground signalling mediates diverse root-root and root-microbe interactions. The Biochemist. 36 (5). 32-3
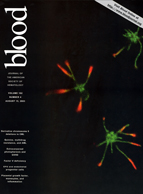
Jiang, J., Woulfe, D. S., Papoutsakis, E. T. 2014. Shear enhances thrombopoiesis and formation of microparticles that induce megakaryocytic differentiation of stem cells. Blood. 124 (13): 2094-2103.
Cover Photo: Biomechanical and notably shear forces are essential for megakaryocytic maturation and platelet biogenesis, but also for generating megakaryocytic microparticles, which are capable of programming hematopoietic stem and progenitor cells to differentiate into megakaryocytes. This immunofluorescent image shows a partially disintegrated human polyploid megakaryocyte giving rise to various megakaryocytic particles through β1 tubulin (green) extensions. F-actin is shown in red and the nuclear structures are shown in blue (TO-PRO-3).

Pradhan-Bhatt, S., Harrington, D. A., Duncan, R. L., Jia, X., Witt, R. L., Farach-Carson, M. L. 2013. Implantable three-dimensional salivary spheroid assemblies demonstrate fluid and protein secretory responses to neurotransmitters. Tissue Engineering Part A. 19 (13-14): DOI: 10.1089/ten.tea.2012.0301
Cover Photo: Alpha-amylase release (red) upon treatment with 50 μm norepinephrine for 60 min

Pradhan-Bhatt, S., Harrington, D. A., Duncan, R. L., Jia, X., Witt, R. L., Farach-Carson, M. L. 2013. Implantable three-dimensional salivary spheroid assemblies demonstrate fluid and protein secretory responses to neurotransmitters. Tissue Engineering Part A. 19 (13-14): DOI: 10.1089/ten.tea.2012.0301
Cover Photo: Merging salivary acini-like structures stained with beta-catenin (green)

Pradhan-Bhatt, S., Harrington, D. A., Duncan, R. L., Jia, X., Witt, R. L., Farach-Carson, M. L. 2013. Implantable three-dimensional salivary spheroid assemblies demonstrate fluid and protein secretory responses to neurotransmitters. Tissue Engineering Part A. 19 (13-14): DOI: 10.1089/ten.tea.2012.0301
Cover Photo: Three-dimensional surface reconstruction of primary human salivary acinar-derived cells in hyaluronic acid-based hydrogels. Individual cells form aggregate clusters, which gradually expand, develop a lumen, and respond to neurotransmitters. Cells in the image were stained with live/dead (green/red) staining and blue nuclei, and imaged by confocal microscopy. The resulting z-stack was reconstructed in Amira software.

Wagner, R., Modla, S., Hossler, F., Czymmek, K. 2012. Three-dimensional analysis and computer modeling of the capillary endothelial vesicular system with electron tomography. Microcirculation. 19 (6): 477-484.

Wei, D., Jacobs, S., Modla, S., Zhang, S., Young, C.L., Cirino, R., Caplan, J., Czymmek, K. 2012. High-resolution three-dimensional reconstruction of a whole yeast cell using focused-ion beam scanning electron microscopy. Biotechniques. 53 (1):41–48.
Cover Photo: Focused-ion beam scanning electron microscopy and 3D segmentation of ER (yellow), mitochondria (red) and nucleus (blue) of a whole yeast cell at 5nm isotropic voxels.

Knapp, E., Flores, R., Scheiblen, D., Modla, S., Czymmek, K., Yusibov, V. 2012. A cryohistological protocol for preparation of large plant tissue sections for screening intracellular fluorescent protein expression. Biotechniques. 52 (1): 31-37.
Cover Photo: Three-dimensional confocal microscopy maximum intensity projection of plant cell wall (blue), chloroplasts (red) and GFP expression (green) in sucrose-infiltrated N. benthamiana leaves agroinfiltrated with the movement-competent TMV expression vector D4GFP at 12 dpi. Image courtesy of E. Knapp, R. Flores, D. Scheiblin, S. Modla, K. Czymmek, and V. Yusibov. Design by Yusef Ramelize.

Bowes, B. D., Traylor, S. J., Timmick, S. M., Czymmek, K. J., Lenhoff, A. M. 2012. Insights into protein sorption and desorption on dextran-modified ion-exchnage media. Chem. Eng. Technol. 35 (1): 91-101.
Cover Photo: Top left: polarization anisotropy map for fluorescently labeled lysozyme in solution and adsorbed on SP Sepharose XL particles. Top right (courtesy of Dr. Harun Koku): SEM of Sepharose FF (left half) and Sepharose XL (right half), both with protein adsorbed. Bottom right: photobleaching of fluorescently labeled lysozyme adsorbed on SP Sepharose FF. Bottom left: confocal microscopy of simultaneous loading of fluorescently labeled lysozyme (green) and lactoferrin (red) on SP Sepharose FF.
Naik, M. U., Stalker, T. J., Brass, L. F., Naik, U. P. 2012. JAM-A protects from thrombosis by suppressing integrin αIIbβ3-dependent outside-in signaling in platelets. Blood. 119 (14): 3352-3360.
Cover Photo: The image is a histological micrograph (magnification ×200) of a lung section of a Jam-A knockout (Jam-Agt/gt) mouse showing both macro- and micro-thromboemboli. Pulmonary thromboembolism was induced by injecting a mixture of collagen and epinephrine through the tail vein.
Wagner, R., Van Loo, D., Hossler, F., Czymmek, K., Pauwels, E., Hoorebeke, L. 2011. High-resolution imaging of kidney vascular corrosion casts with Nano-CT. Microscopy and Microanalysis. 17 (2): 215-219.
Cover Photo: Nano-CT image of a vascular corrosion cast of an entire mouse kidney.

Hillwig M.S., Lebrasseur N.D., Green P.J., Macintosh G.C. 2008. Impact of transcriptional, ABA-dependent, and ABA-independent pathways on wounding regulation of RNS1 expression. Mol. Genet. Genomics. 280 (3):249-61.

Hoiczyk, E., Ring, M. W., McHugh, C. A., Schwär, G., Bode, E., Krug, D., Alrmeyer, M. O., Lu, J. Z., Bode, H. B. 2009. Lipid body formation plays a central role in cell fate determination during develomental differentiation ofMyxococcus xanthus. Mol. Microbiol. 74 (2): 497-517.
Cover Photo: A transmission electron micrograph of a high-pressure frozen complete fruiting body of Myxococcus xanthus after 48h is shown at the top, in which the individual spores are visible as red-colored structures throughout the mound. On the lower left, isolated 48h old spores have been stained with the lipid-specific fluorescent dye nile red, showing that each spore contains multiple lipid bodies that are clearly visible at high resolution in the electron micrograph on the right.

Oristian, D.S., Sloofman, L.G., Zhou, Z., Wang, L., Farach-Carson, M.C. and Catherine B. Kirn-Safran. 2009. Ribosomal protein L29/HIP deficiency delays osteogenesis and increases fragiligy of adult bone in mice. J Orthopædic Res. 27(1): 28-35.
Cover Photo:Immunostaining for early and late markers of chondrocytedifferentiation on consecutive sections: type II (A, B) and type X collagen (C, D). Arrows (C, D) delineate the length of hypertrophic zones. Arrowheads in (A) indicate the periphery of developing bone.

Patrick Ollivier, Andrew Bahrou, Sarah Marcus, Talisha Cox, Thomas Church and Thomas Hanson. 2008. Volatilization and Precipitation of Tellurium by Aerobic, Tellurite Resistant Marine Microbes. Appl. Environ. Microbiol. 74(23):7163-7173.
Cover Photo: Thin-section transmission electron micrograph (TEM) of Bacillus sp. strain 6A with precipitated tellurium nanostructures highlighted in light blue. Cell constituents are colored deep blue and green on an orange field. The original TEM image was viewed using a Zeiss CEM 902 at 80 kV and captured with a Soft Imaging System Mega View II camera. The image was false colored using Adobe Photoshop CS3. Photo courtesy of Shannon Modla.
Thimmaraju Rudrappa, Kirk J. Czymmek, Paul W. Par and Harsh P. Bais. 2008. Root-Secreted Malic Acid Recruits Beneficial Soil Bacteria. Plant Physiology. 148(3):1547–1556.
Cover Photo: A confocal three-dimensional maximum intensity projection of Bacillus subtilis biofilm formation (green) on living Arabidopsis thaliana Col-0 root stained with Calcoflour White (blue). Bacteria and plant nuclei (green) stained green with vital nucleic acid stain Syto 13.

Linda Sequeira, Cara Dubyk, Tracy Riesenberger, Carlton Cooper and Kenneth Van Golen. 2008. Rho GTPases in PC-3 prostate cancer cell morphology, invasion and tumor cell diapedesis. Clin Exp Metastasis (2008) 25:569–579.
Cover Photo: Effect of Rho and Rac GTPases on BMEC binding and diapedesis. 3-D composite images of diapedesis of PC-3 cells transfected with either shScr control or shRac. The shScr images were taken 45 min and the shRac at 60 min after adding the tumor cells to the BMEC layer.

Xiaochun Zhang and, Andrew V. Teplyakov. 2008. Adsorption of C60Buckminster Fullerenes on an 11-Amino-1-undecene-Covered Si(111) Substrate. Langmuir. 24(3):810-820.
Cover Photo: An illustration collected and created by Xiaochun Zhang presents the atomic force microscopy images of fullerene buckyball C60 molecules attached to the mixed 11-amino-1-undecence and decene self-assembled monolayers (SAMS) on Si(111)substrate.

Kirk Czymmek, Melissa Fogg, Deborah Powell, James Sweigard, Sook-Young Park, Seogchan Kang. 2007. In vivo time-lapse documentation using confocal and multi-photon microscopy reveals the mechanisms of invasion into the Arabidopsis root vascular system by Fusarium oxysporum. Fungal Genetics & Biology. 44:1011-1023.
Cover Photo: Reef coral fluorescent protein ZsGreen labeled Fusarium oxysporum (green) infection of the lateral root (red) of Arabidopsis susceptible ecotype Cape Verdi Island (Cvi-0) taken 10 days post-inoculation. This three-dimensional shadow projection was created from 37 optical slices acquired at a 1 micron z-interval.

Veeco. Sample courtesy of Liz Adams and Randall Duncan. Microscopy and Analysis, The Americas, March 2007, 83.
Cover Photo: A network of actin fibers that make up the cytoskeleton of live MC3T3F osteoblast cells which are mononucleated cells responsible for bone formation. Cells were imaged on a Veeco Bioscope II atomic force microscope (AFM). Scan size 100µm.

Manisha Malik and Robert A. Sikes. April 2006. Biotechniques. 40 (4).
Cover Photo: Human bone marrow endothelial-1 (HBME-1) cells. Mitochondria (red) were labeled in living cells with MitoTracker® Red; a biotinylated phage display peptide (L13) was localized with streptavidin-conjugated Alexa Fluor™ 488 (green); and nuclei (blue) were labeled with DRAQ5™. Cells were imaged using a Zeiss 510 NLO VIS confocal microscope.

R.C. Wagner, K. Czymmek and F.E. Hossler. 2006. Confocal Microscopy, Computer Modeling and Qauntification of Glomerular Vascular Corrosion Casts. Microscopy and Microanalysis.12:262-268.
Cover Photo: Confocal microscope image of a casted but uncorroded rabbit kidney renal corpuscle. Red regions highlight capillaries and arterioles, while green regions are stained to show various cell nuclei. Full width = 188um.

Ronald R. Gomes Jr., Mary C. Farach-Carson, and Daniel D. Carson. 2004. Perlecan Functions in Chondrogenesis: Insights from in vitro and in vivo Models. Cells Tissues Organs. 176:79-86.
Cover Photo: P/n domain I-stimulated mesenchymal cess condensation and differentiation.
Stephanie A. Miller, Anissa J. Brown, Mary C. Farach-Carson, and Catherine B. Kirn-Safran. 2003. HIP/RPL29 down-regulation accompanies terminal chondrocyte differentiation. Differentiation. 71:322-336.
Cover Photo: Confocal image of the developing murine cartilage growth plate stained for perlecan (green), F-actin (red) and nuclei (blue).
Naik, U. P. and Meghna, U. N. 2003. Association of CIB with GPIIb/IIIa during outside-in signaling is required for platelet spreading on fibrinogen. Blood. 102 (4): 1355-1362.
Cover Photo: Localization of the CIB (red) and GPIIb (green) in a spiky, partially spread platelet.

S.R. Dziennik, E.B. Belcher, G.A. Barker, M.J. DeBergalis, S.E. Fernandez, and A.M. Lenhoff. 2003. Nondiffusive mechanisms enhance protein uptake rates in ion exchange particles. PNAS. 100:420-425. Copyright 2003 National Academy of Sciences, U.S.A.
Cover Photo: Laser scanning confocal microscopic image of fluorescently labeled hen egg lysozyme in cation exchange particles. The bright red rings indicate areas of higher protein concentration and occur only after initial protein uptake. These findings demonstrate that mechanisms other than diffusion contribute to protein transport in oppositely charged porous material.

Timothy M. Bourett, James A. Sweigard, Kirk J. Czymmek, Anne Carroll and Richard J. Howard. 2002. Reef coral fluorescent proteins for visualizing fungal pathogens. Fungal Genetics & Biology. 37:211-220. Copyright 2002, with permission from Elsevier.
Cover Photo: A spectral fingerprint-based image of RCFP-expressing live cells. A Zeiss META system was employed to differentiate between germinating conidia of F. verticilloides expressing AmCyan (blue), ZsGreen (green), and ZsYellow (red).
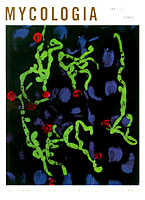
Kirk J. Czymmek, Timothy M. Bourett, James A. Sweigard, Anne Carroll and Richard J. Howard. 2002. Utility of cytoplasmic fluorescent proteins for live-cell imaging of Magnaporthe grisea in planta. Mycologia. 94:280-289.
Cover Photo: Cytoplasmic expression of green fluorescent protein in Magnaporthe grisea (green) infecting barley leaf (blue), imaged by Multiphoton microscopy. Appressoria (red) exhibited strong signal after pulsed, near infrared laser excitation false color, maximum intensity projection of thirty optical sections.

Michael C. Rice, Michael Bruner, Kirk Czymmek and Eric B. Kmiec. 2001. In vitro and in vivo nucleotide exchange directed by chimeric RNA/DNA oligonucleotides in Saccharomyces cerevisae. Molecular Microbiology. 40:857-868.
Cover Photo: Gene repair of a poit mutation located within a fusion reporter gene construct directed within a fusion reporter gene construct directed by synthetic oligonucleotides in Saccharomyces cerevisiae. Correction enables the normal protein to bind a ligand and emit green fluorescence.

Timothy M. Bourett, Kirk J. Czymmek and Richard J. Howard. 1998. An Improved Method for Affinity Probe Localization in Whole Cells of Filamentous Fungi. Fungal Genetics & Biology. 24:3-13. Copyright 1998, with permission from Elsevier.
Cover Photo: Cluster of nine mitotic nuclei in a hyphal cell of Rhizoctonia oryzae prepared using a novel, cryo-based procedure. The whole intact cell was probed with an α-tubulin antibody (red fluorescence) and the nucleic acid-binding dye Syto13 (green fluorescence). Regions of overlapping fluorescence appear yellow. Thick bundles of spindle microtubules as well as astral microtubules are visible in this maximum intensity projection of laser-scanning confocal optical sections.
